 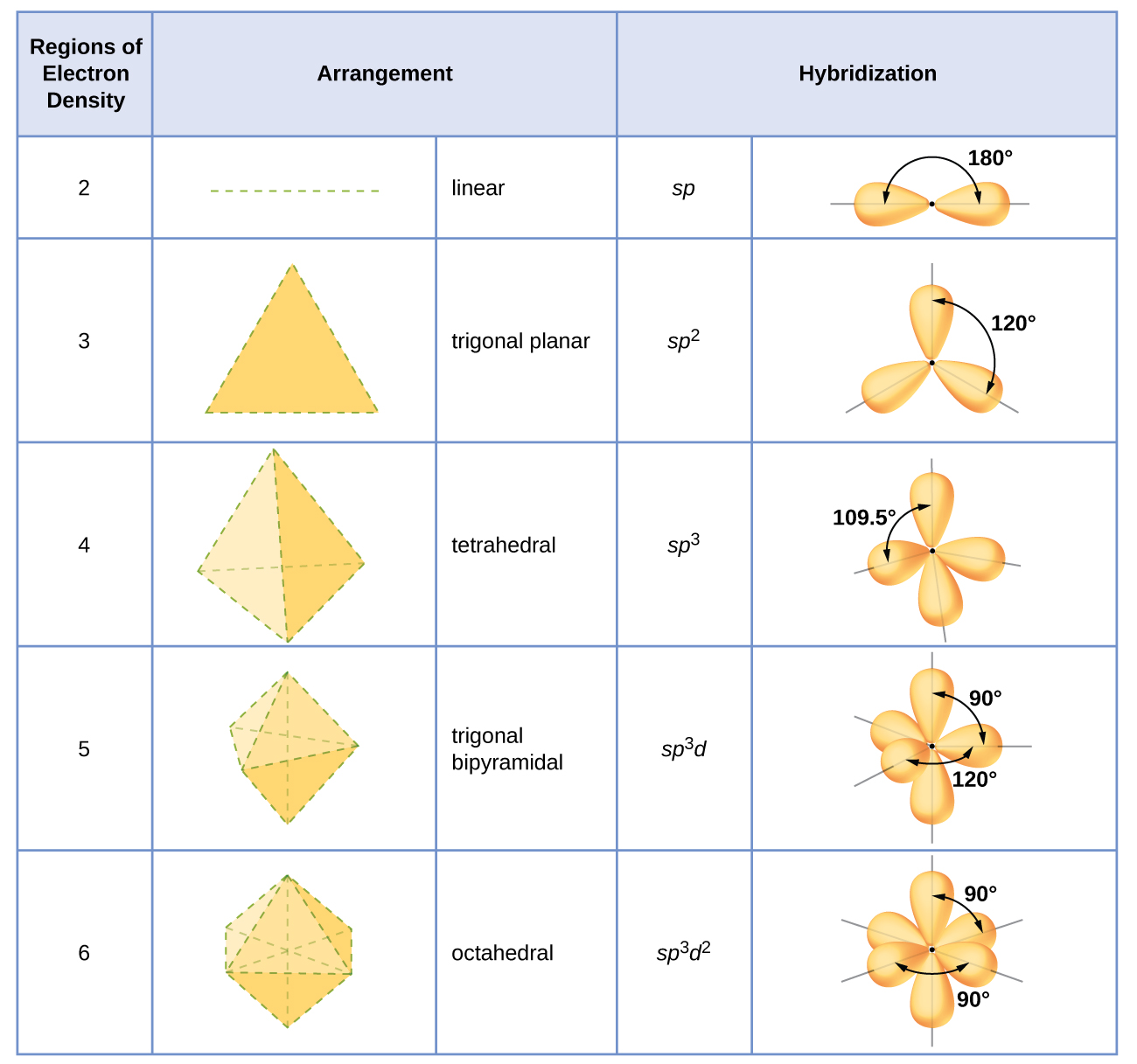
For the geometry, we have another theory called VSEPR theory. Lewis structure does not attempt to predict molecular geometry and shape. Here is a video attached regarding steps to draw the lewis structure of AlCl3. Thus, the structure drawn in step 6 is the best Lewis structure for AlCl3. Total number of valence electrons in a free atom Therefore, the sum of formal charge on three atoms should come out to be zero. Calculate the formal charge on all atoms. It is always short of two electrons, and hence it is an electron-deficient compound. There is no alternate lewis structure in which the octet is complete. It shares one electron from all Cl-atoms, and still, the octet does not go to completion. 
They share one electron with Al to have a fully filled valence shell configuration.Īl has three valence electrons in the isolated state. Complete the octet of atoms by forming bonds.Įach Cl has seven valence electrons in the isolated state. The blue dots represent electrons from Al, and black dots represent electrons from Cl. The total valence shell electrons (calculated in step 1) are placed according to a predicted bond formation. 
Arrange the valence electrons around the elemental symbols. In this step, we have to arrange the side atoms and central atoms suitably. Thus, Al is the central atom for this compound. The central atom is supposed to be the least electronegative one out of the constituent atoms as the central atom is supposed to share its electron density with all other atoms. Choose a suitable central atom for the compound. The Lewis dot structure for Al and Cl are as follows. The chemical symbols for aluminum and chlorine are Al and Cl, respectively. We draw the Lewis structure of elements by arranging the valence shell electrons around the element’s chemical symbol. Draw the lewis dot structure for elements. Total number of valence shell electrons= 3 + (7*3) = 24 Valence electrons according to group number Count the total number of valence shell electrons on the compound.īefore drawing the structure, we need to know the number of valence shell electrons on all constituent atoms and their sum. It is calculated using the given formula-įormal charge= (number of electrons in an isolated neutral atom)- (number of nonbonding electrons on an atom in the compound)- 0.5*(number of electrons shared in bonds by atom) It compares the number of valence electrons in an isolated neutral atom with the number of valence electrons in the bonded form in a molecule. This preference for 8 electrons gives rise to the octet rule. Hence all main group elements prefer to have a fully filled configuration by having a net of 8 electrons in their valence shell after bonding. Noble gases have eight electrons in their valence shell, except He. Every element forms a bond to attain a noble gas-like configuration. Noble gases are considered the least reactive and most stable in the periodic table. To find the most accurate and lowest energy structure, we must satisfy the octet rule and formal charges on all the constituent atoms. There can be a possibility of more than one Lewis structure for some compounds. In this method, valence shell electrons are represented by dots on the chemical symbol of elements. He postulated that only valence electrons take part in bonding, and hence in his representation, only valence electrons are considered. Lewis was among the first ones to explain. Many attempts were made to explain the formation of the chemical bond. Lewis Structure is a 2-D representation that depicts the arrangement of atoms and valence shell electrons on those atoms in a compound. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
